Innovative Clinical Solutions in Biotechnology

Transforming lives through innovation in clinical sciences
Summary
Performance-driven, resilient, and adaptable pharmaceutical and biotechnology executive with accomplishments built upon visionary and strategic acumen, collaborative empowerment, and service centered leadership. Deep expertise in disease mechanisms, progression, and treatment in neurology, oncology, immunology, infectious diseases and dermatology with small molecules, biomolecules and gene therapy. Proven track record in integrating this knowledge to design and execute complex clinical trials, optimize endpoint strategies, and achieve regulatory approvals.
Leadership
Rebuilt Chief Medical office, regulatory and clinical development infrastructure for more than 18 biotechnology companies to transform regulatory, financial and operational strategies.

Service centered leadership style and people management (including hiring, firing, line budget authority) in medical, clinical, chemical and engineering professions (MD/PhD, PhD, BioMed Engineers, Mech.E, EE) developing novel indications in neurology, oncology and immunology.
Financial Stewardship

Stewardship of capital in biotechnology companies is critical to achieving marketing approval partnerships. I have successfully acquired more than $200MM funding from NIH, DoD, BARDA for clinical development in oncology and neurology.
Extensive scientific, engineering, clinical, medical and regulatory management experience of large and complex neurology clinical development programs with academic clinical centers, alliance partners, HHS and DoD/MoD agencies helped build programs with budgets ranging from $20MM through $80MM.
Therapeutic Areas and Populations

Therapeutic area expertise span immunology, oncology, neurology, hematology, and dermatology to drive impactful clinical development and regulatory success built from deep expertise in disease mechanisms and progression for ultra-rare, rare and non-rare diseases
Identify and Implement Optimal Route of Administration for Target Therapeutic Areas

Clinical development expertise enabled Phase 1-4 trials for small molecules, biologicals, drug/device combinations, cell and gene therapies spanning multiple routes of drug delivery including oral, pulmonary, parenteral and drug/device combinations. Led design and execution of both AAV and Lentiviral (HSCT) clinical trials.
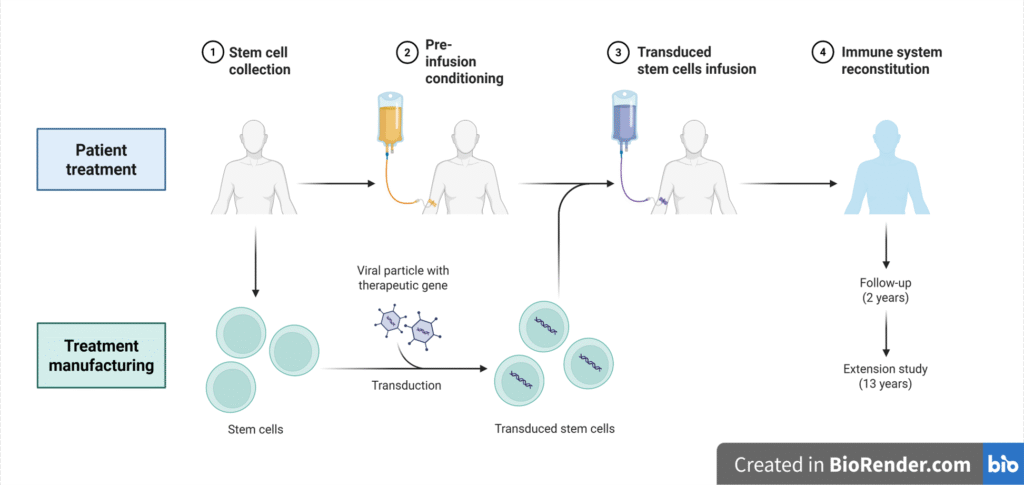
Inter and Intra-Institutional Leadership

Led cross-institutional negotiations between DoD, BARDA, FDA and NIH with industry partners to advance drug-device combinations through marketing approval in emergent CNS diseases.
Delivered NIH funded registry studies to support marketing approval of novel indications in CNS, surgical reconstruction and progressive neurodegenerative diseases.

Provided scientific, engineering, clinical, medical and regulatory management experience of large and complex neurology clinical development programs with academic clinical centers, alliance partners, HHS and DoD/MoD agencies with program budgets ranging from $20MM through $80MM.
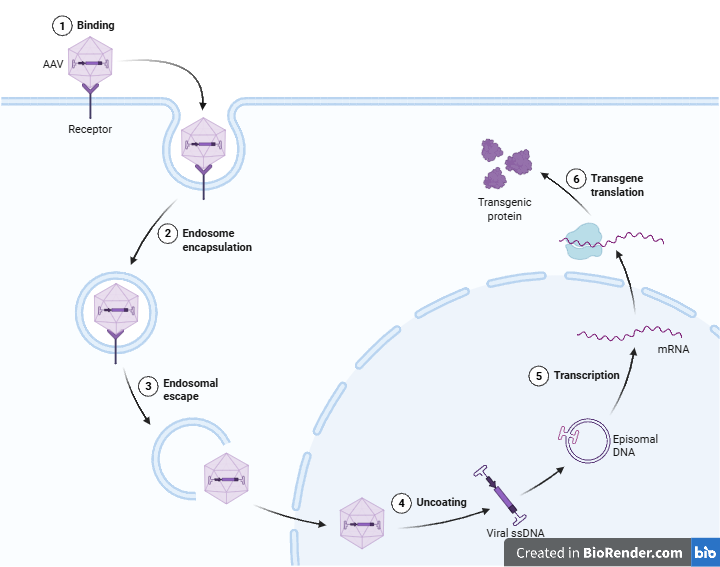
Led gene and cell therapy clinical development in six biotechnology, venture capital funded companies from preclinical studies through registrational stage.

Led Chief Medical office, regulatory and clinical development infrastructure for more than 18 biotechnology companies that realized regulatory, financial and operational risks.

Service centered leadership style and people management (including hiring, firing, line budget authority) in medical, clinical, chemical and engineering professions (MD/PhD, PhD, BioMed Engineers, Mech.E, EE) developing novel indications in neurology, oncology and immunology.
Artificial Intelligence & Machine Learning

Experienced in leveraging AI/ML to enhance pharmaceutical innovations and clinical operations and statistical methodologies, aimed at advancing therapeutic solutions across various disease areas while maintaining a focus on regulatory strategy and patient outcomes and across gene therapy, immunology, oncology, neurology, hematology, and dermatology.
AI/ML Trial Design
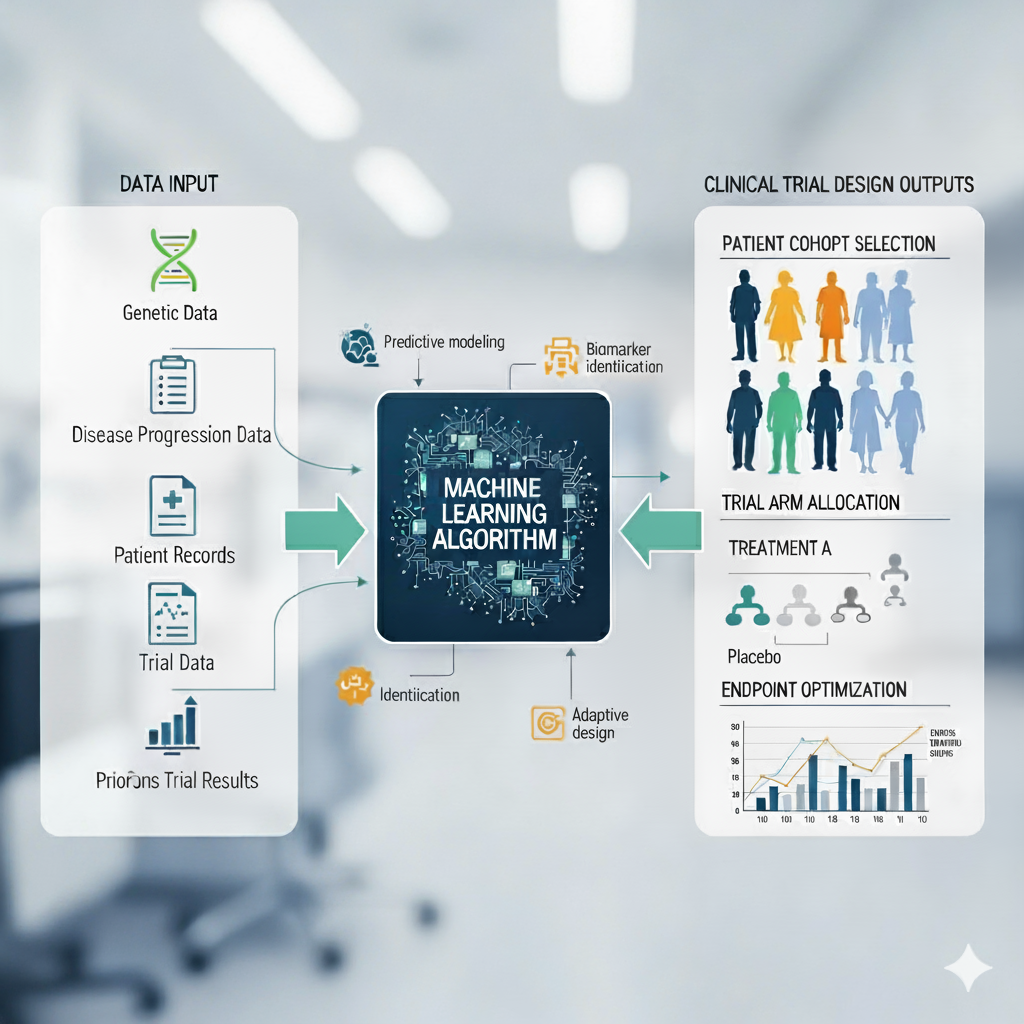
Leveraging deep expertise in disease mechanisms augmented with machine learning (ML: R, SAS, Python) founded upon treatment strategies to design and execute complex clinical trials, ensuring optimal engagement with medical institutions for timely regulatory approvals.
Pharmaceutical Industry Expertise
Leverage my extensive experience in clinical development to drive innovation in gene therapy, immunology, oncology, neurology, hematology, and dermatology.